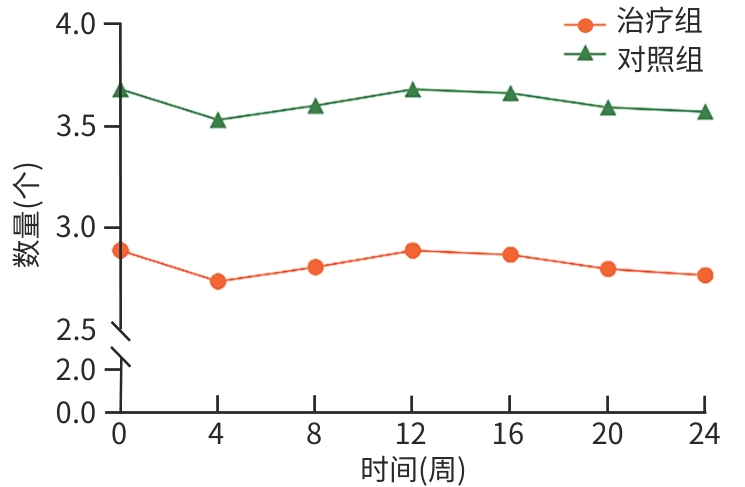
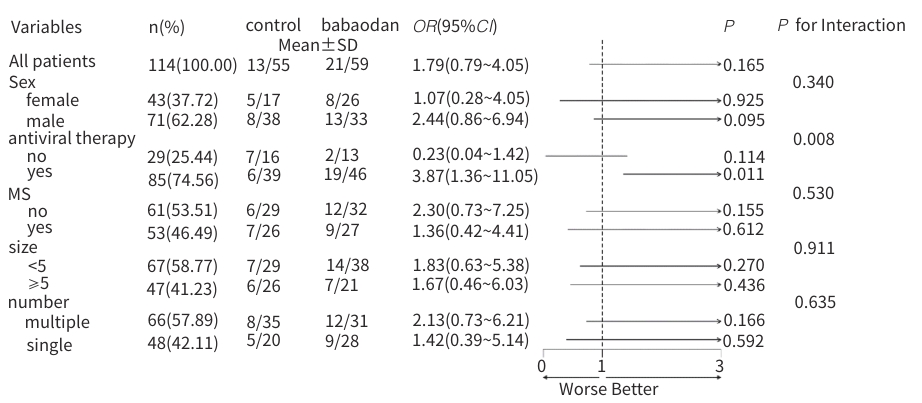
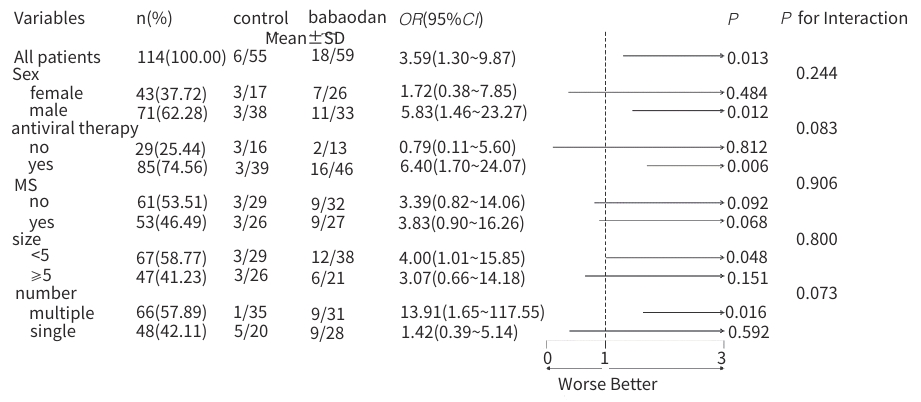
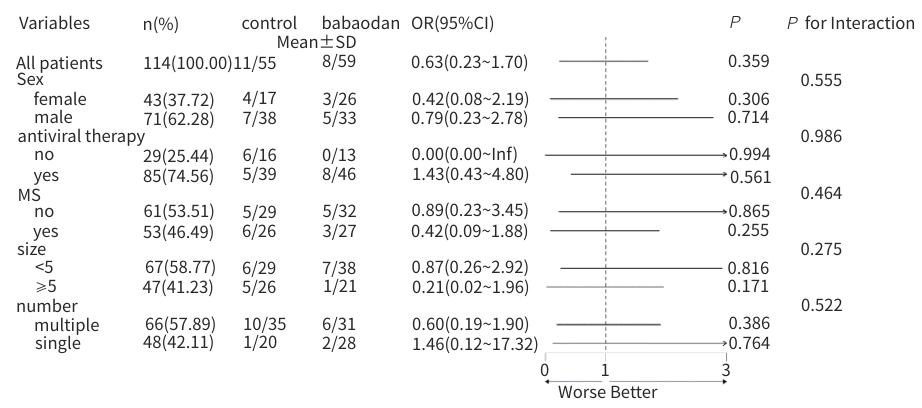
| [1] |
FOLEY KG, LAHAYE MJ, THOENI RF, et al. Management and follow-up of gallbladder polyps: Updated joint guidelines between the ESGAR, EAES, EFISDS and ESGE[J]. Eur Radiol, 2022, 32( 5): 3358- 3368. DOI: 10.1007/s00330-021-08384-w. |
| [2] |
ZHENG YM, BAI XS, YAO GB, et al. Risk factors of gallbladder polyps formation in East Asian population: A meta-analysis and systematic review[J]. Asian J Surg, 2020, 43( 1): 52- 59. DOI: 10.1016/j.asjsur.2019.03.015. |
| [3] |
MAO YS, MAI YF, LI FJ, et al. Prevalence and risk factors of gallbladder polypoid lesions in Chinese petrochemical employees[J]. World J Gastroenterol, 2013, 19( 27): 4393- 4399. DOI: 10.3748/wjg.v19.i27.4393. |
| [4] |
CHEN SP, WANG ZX, ZHANG XD, et al. Epidemiology of polypoid lesions of the gallbladder and related risk factors[J]. J Clin Hepatol, 2019, 35( 2): 441- 443. DOI: 10.3969/j.issn.1001-5256.2019.02.045. |
| [5] |
HUANG TY, ZHANG RH, LUO H. Study on the therapeutic effect of single-port and multi-port laparoscopic surgery on benign gallbladder polyps[J]. Trauma and Crit Medicine, 2024, 12( 4): 245- 247. DOI: 10.16048/j.issn.2095-5561.2024.04.11. |
| [6] |
YANG HX, GOU J, HUANG P, et al. Babaodan Capsules for viral hepatitis: Systematic review of clinical efficacy and safety and Meta-analysis of randomized clinical trials[J]. China J Chin Mater Med, 2019, 44( 22): 4953- 4961. DOI: 10.19540/j.cnki.cjcmm.20190627.501. |
| [7] |
LIU PF. Clinical effect of Babaodan capsules in the treatment of jaundice type viral hepatitis[J]. Clin Res Pract, 2018, 3( 10): 138- 139. DOI: 10.19347/j.cnki.2096-1413.201810066. |
| [8] |
|
| [9] |
Hepatobiliary Disease Branch of Chinese Society of Traditional Chinese Medicine. Expert consensus on clinical application of Babaodan capsule in the treatment of viral hepatitis[J]. Chin J Integr Tradit West Med Liver Dis, 2020, 30( 6): 579- 580.
中华中医药学会肝胆病学分会. 八宝丹胶囊治疗病毒性肝炎临床应用专家共识[J]. 中西医结合肝病杂志, 2020, 30( 6): 579- 580.
|
| [10] |
REN JB, SHEN YX, ZHANG YG, et al. Clinical trial of Babaodan capsules in the treatment of patients with acute cholecystitis[J]. Chin J Clin Pharmacol, 2023, 39( 12): 1689- 1693. DOI: 10.13699/j.cnki.1001-6821.2023.12.004. |
| [11] |
Spleen and Stomach Disease Branch of Chinese Society of Traditional Chinese Medicine. Consensus opinion of TCM diagnosis and treatment experts on cholecystitis(2017)[J]. Chin J Integr Tradit West Med Dig, 2017, 25( 4): 241- 246. DOI: 10.3969/j.issn.1671-038X.2017.04.01. |
| [12] |
Hepatobiliary Specialized Committee of China Association of Chinese Medicine, Liver Diseases Specialized Committee of China Medical Association of Minorities. The clinical guidelines of diagnosis and treatment of chronic hepatitis B with traditional Chinese medicine(2018)[J]. J Clin Hepatol, 2018, 34( 12): 2520- 2525. DOI: 10.3969/j.issn.1001-5256.2018.12.007.
|
| [13] |
SHAO H. Clinical observation on 68 cases of polypoid lesions of gallbladder treated with self-made Huayu Lidan decoction[J]. Forum Tradit Chin Med, 2004, 19( 2): 33- 34. DOI: 10.3969/j.issn.1002-1078.2004.02.027. |
| [14] |
JIANG H, CHEN HQ. Clinical study on Xiaoyan Lidan Tang for gallbladder polyps[J]. J New Chin Med, 2019, 51( 6): 172- 174. DOI: 10.13457/j.cnki.jncm.2019.06.051. |
| [15] |
YUAN SQ. Study on the clinical efficacy of Shiwuwei Gandanshu Pill in the treatment of polypoid lesions of gallbladder(damp-heat of liver and gallbladder with blood stasis syndrome)[J]. Zhengzhou: Henan University of Chinese Medicine, 2018.
袁帅强. 十五味肝胆舒丸治疗胆囊息肉样病变(肝胆湿热夹血瘀证)临床疗效研究[D]. 郑州: 河南中医药大学, 2018.
|
| [16] |
LI RJ, LI ZQ, DANG ZQ, et al. Self-made Lidan Hewei Granules combined with Danshi Tongli Tablets in the treatment of 40 cases of gallbladder polyps[J]. Chin J Ethnomed Ethnopharmacy, 2016, 25( 17): 97- 98, 100. DOI: 10.3969/j.issn.1007-8517.2016.17.zgmzmjyyzz201617035. |
| [17] |
LI YF, SHENG HD, QIAN J, et al. The Chinese medicine babaodan suppresses LPS-induced sepsis by inhibiting NLRP3-mediated inflammasome activation[J]. J Ethnopharmacol, 2022, 292: 115205. DOI: 10.1016/j.jep.2022.115205. |
| [18] |
QIAN J, XU HD, LV DQ, et al. Babaodan controls excessive immune responses and may represent a cytokine-targeted agent suitable for COVID-19 treatment[J]. Biomed Pharmacother, 2021, 139: 111586. DOI: 10.1016/j.biopha.2021.111586. |
| [19] |
LI GD, LI XP, FENG CY, et al. Comparative study on the effects of in vitro cultured bezoar and natural bezoar on the metabolic profiles of serum and tissue bile acids in mice[J]. Lishizhen Med Mater Med Res, 2021, 32( 6): 1328- 1331. DOI: 10.3969/j.issn.1008-0805.2021.06.13. |
| [20] |
SONG J, LI J, GUO SJ, et al. An initial observation about the therapeutic effect of Danweishu& yunjingyi treatment on gallbladder polyps[J]. Chin Arch Tradit Chin Med, 2010, 28( 1): 168- 170. DOI: 10.13193/j.archtcm.2010.01.170.songj.021. |
| [21] |
SHENG DD, ZHAO SM, GAO L, et al. BabaoDan attenuates high-fat diet-induced non-alcoholic fatty liver disease via activation of AMPK signaling[J]. Cell Biosci, 2019, 9: 77. DOI: 10.1186/s13578-019-0339-2. |







 DownLoad:
DownLoad: