肠道微生物在慢性胰腺炎胰腺纤维化中的作用机制及相关治疗策略
DOI: 10.12449/JCH260233
利益冲突声明:本文不存在任何利益冲突。
作者贡献声明:颜运君负责设计论文框架,起草论文;盛亮、王祺负责查找资料、绘制图表;彭顺、李佳负责拟定写作思路;张磊指导撰写文章并最后定稿。
Mechanism of action of gut microbiota in chronic pancreatitis fibrosis and related treatment strategies
-
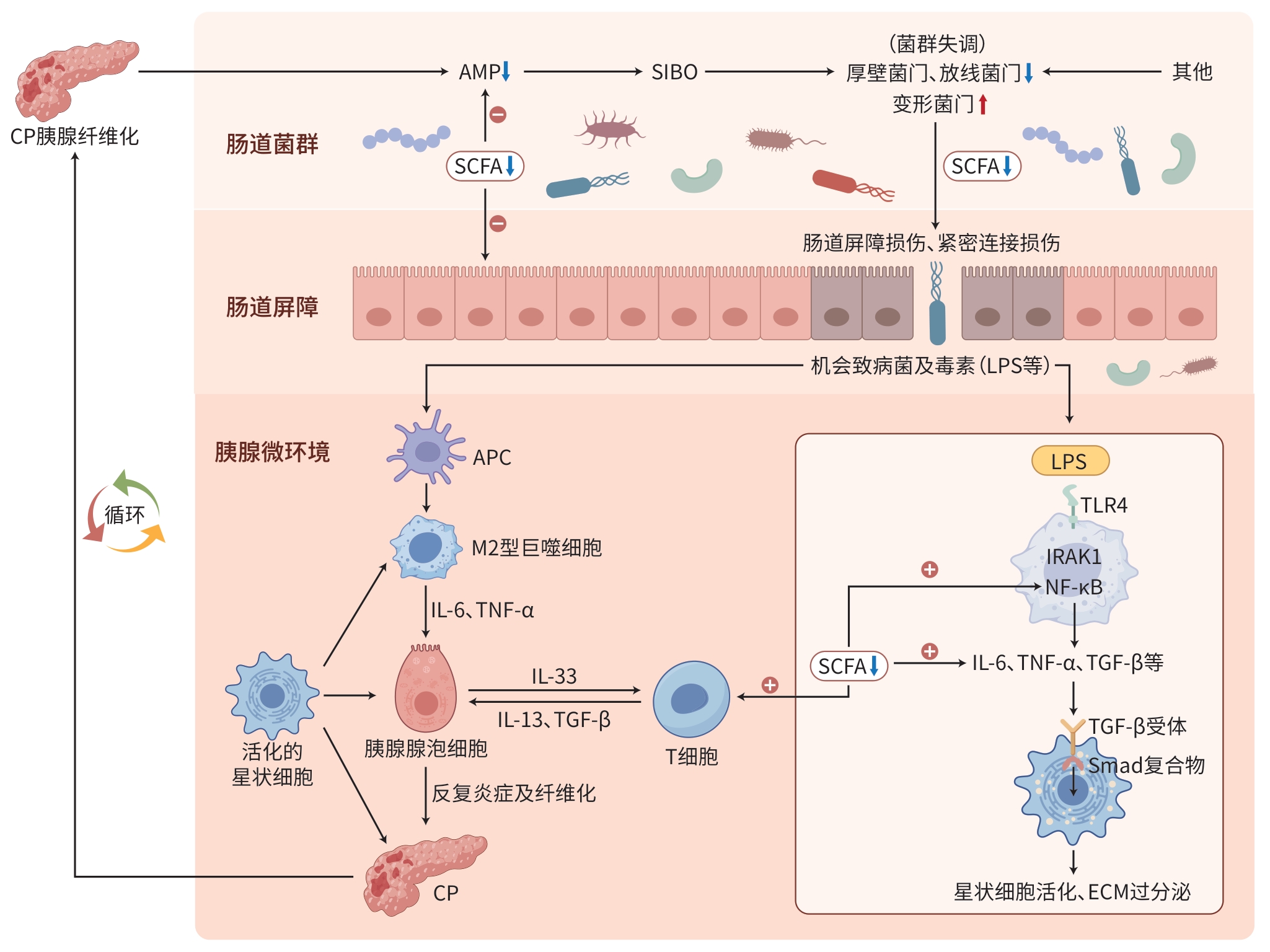
摘要: 慢性胰腺炎(CP)是以胰腺进行性炎症纤维化为核心病理特征的临床常见疾病。肠道菌群作为“人类第二基因组”,可通过肠-胰轴双向调控CP胰腺纤维化进程。本文系统阐述CP进程中肠道菌群的特征及其通过细菌易位、代谢产物、免疫调控网络及微生物-胰腺星状细胞交互作用介导胰腺纤维化的分子机制,重点剖析短链脂肪酸、炎症因子网络在胰腺星状细胞活化及细胞外基质沉积中的核心作用。同时,探讨益生菌、益生元和粪菌移植等靶向肠道微生态干预策略在防治CP胰腺纤维化中的潜在价值,并展望通过多组学技术筛选CP诊断标志物和新型治疗靶点的转化前景,旨在为CP的精准诊疗提供新思路。Abstract: Chronic pancreatitis (CP) is a common disease in clinical practice characterized by progressive inflammatory fibrosis of the pancreas. Gut microbiota, known as the “second genome” of humans, bidirectionally modulates the progression of fibrosis in CP via the gut-pancreas axis. This article systematically elaborates on the characteristics of gut microbiota during the progression of CP and its molecular mechanism in mediating pancreatic fibrosis through bacterial translocation, metabolites, immune regulatory networks, and microbe-pancreatic stellate cell interactions, with a focus on the pivotal role of short-chain fatty acids and inflammatory cytokine networks in pancreatic stellate cell activation and extracellular matrix deposition. In addition, this article explores the potential value of gut microbiota-targeted interventions in the prevention and treatment of CP fibrosis, such as probiotics, prebiotics, and fecal microbiota transplantation, and discusses the translational potential of using multi-omics technologies to identify diagnostic biomarkers and novel therapeutic targets for CP, in order to provide new ideas for the precise diagnosis and treatment of CP.
-
Key words:
- Gastrointestinal Microbiome /
- Pancreatitis, Chronic /
- Fibrosi /
- Therapeutics
-
[1] HAN C, WANG D, HU LH, et al. Current status of prospective cohort studies on chronic pancreatitis[J]. Chin J Pancreatol, 2021, 21( 3): 161- 167. DOI: 10.3760/cma.j.cn115667-20201222-00209.韩超, 王丹, 胡良皞, 等. 慢性胰腺炎前瞻性队列研究现状[J]. 中华胰腺病杂志, 2021, 21( 3): 161- 167. DOI: 10.3760/cma.j.cn115667-20201222-00209. [2] KONG FY, LI ZS. Research progress of pancreatic diseases[J]. Chin J Pancreatol, 2021, 21( 1): 6- 15. DOI: 10.3760/cma.j.cn115667-20201125-00191.孔凡扬, 李兆申. 胰腺疾病的研究进展[J]. 中华胰腺病杂志, 2021, 21( 1): 6- 15. DOI: 10.3760/cma.j.cn115667-20201125-00191. [3] SUN YY, LU YY, JING DD. Research progress of intestinal microbiology in the development of chronic pancreatitis and pancreatic cancer[J]. Int J Dig Dis, 2021, 41( 4): 240- 243. DOI: 10.3969/j.issn.1673-534X.2021.04.003.孙莹莹, 陆颖影, 靖大道. 慢性胰腺炎和胰腺癌发生及发展中肠道微生态的研究进展[J]. 国际消化病杂志, 2021, 41( 4): 240- 243. DOI: 10.3969/j.issn.1673-534X.2021.04.003. [4] THOMAS RM, GHARAIBEH RZ, GAUTHIER J, et al. Intestinal microbiota enhances pancreatic carcinogenesis in preclinical models[J]. Carcinogenesis, 2018, 39( 8): 1068- 1078. DOI: 10.1093/carcin/bgy073. [5] PUSHALKAR S, HUNDEYIN M, DALEY D, et al. The pancreatic cancer microbiome promotes oncogenesis by induction of innate and adaptive immune suppression[J]. Cancer Discov, 2018, 8( 4): 403- 416. DOI: 10.1158/2159-8290.CD-17-1134. [6] SETHI V, KURTOM S, TARIQUE M, et al. Gut microbiota promotes tumor growth in mice by modulating immune response[J]. Gastroenterology, 2018, 155( 1): 33- 37. e 6. DOI: 10.1053/j.gastro.2018.04.001. [7] GELLER LT, BARZILY-ROKNI M, DANINO T, et al. Potential role of intratumor bacteria in mediating tumor resistance to the chemotherapeutic drug gemcitabine[J]. Science, 2017, 357( 6356): 1156- 1160. DOI: 10.1126/science.aah5043. [8] ADOLPH TE, MAYR L, GRABHERR F, et al. Pancreas-microbiota cross talk in health and disease[J]. Annu Rev Nutr, 2019, 39: 249- 266. DOI: 10.1146/annurev-nutr-082018-124306. [9] ZHANG L, WU YN, ZHANG J, et al. The community structure of intestinal bacteria from cirrhosis patients and its influence factors[J]. Chin J Dig Endosc, 2019, 36( 4): 277- 282. DOI: 10.3760/cma.j.issn.1007-5232.2019.04.012.张磊, 吴永娜, 张静, 等. 肝硬化患者肠道细菌群落结构及其影响因素的研究[J]. 中华消化内镜杂志, 2019, 36( 4): 277- 282. DOI: 10.3760/cma.j.issn.1007-5232.2019.04.012. [10] MENG YT, ZHOU CH, XU JJ, et al. Intestinal microecology and chronic pancreatitis[J]. Chin J Pancreatol, 2019, 19( 6): 470- 474. DOI: 10.3760/cma.j.issn.1674-1935.2019.06.020.孟雨亭, 周春华, 徐佳佳, 等. 肠道微生态与慢性胰腺炎[J]. 中华胰腺病杂志, 2019, 19( 6): 470- 474. DOI: 10.3760/cma.j.issn.1674-1935.2019.06.020. [11] JANDHYALA SM, MADHULIKA A, DEEPIKA G, et al. Altered intestinal microbiota in patients with chronic pancreatitis: Implications in diabetes and metabolic abnormalities[J]. Sci Rep, 2017, 7: 43640. DOI: 10.1038/srep43640. [12] KANG C, WANG B, KALIANNAN K, et al. Gut microbiota mediates the protective effects of dietary capsaicin against chronic low-grade inflammation and associated obesity induced by high-fat diet[J]. mBio, 2017, 8( 3): e00470-17. DOI: 10.1128/mBio.00470-17. [13] LI GP, YANG M, ZHOU K, et al. Diversity of duodenal and rectal microbiota in biopsy tissues and luminal contents in healthy volunteers[J]. J Microbiol Biotechnol, 2015, 25( 7): 1136- 1145. DOI: 10.4014/jmb.1412.12047. [14] LI X. Effects of intestinal microbial dysbiosis on chronic pancreatitis[D]. Wuxi: Jiangnan University, 2019.李璇. 肠道菌群紊乱对慢性胰腺炎的影响及机制研究[D]. 无锡: 江南大学, 2019. [15] JIANG SN, SUN TT, ZHI WB, et al. The role of intestinal microecological imbalance in the development of chronic pancreatitis and the application progress of corrective measures[J]. Shandong Med J, 2024, 64( 33): 102- 106. DOI: 10.3969/j.issn.1002-266X.2024.33.025.姜盛楠, 孙婷婷, 支文冰, 等. 慢性胰腺炎发展中肠道微生态失衡的作用及纠正措施应用进展[J]. 山东医药, 2024, 64( 33): 102- 106. DOI: 10.3969/j.issn.1002-266X.2024.33.025. [16] YAN LH, MU B. Small intestinal bacterial overgrowth and diabetes mellitus[J]. World Chin J Dig, 2017, 25( 26): 2380- 2386. DOI: 10.11569/wcjd.v25.i26.2380.闫丽辉, 穆标. 小肠细菌过生长与糖尿病相关性的研究进展[J]. 世界华人消化杂志, 2017, 25( 26): 2380- 2386. DOI: 10.11569/wcjd.v25.i26.2380. [17] DIMAGNO MJ, FORSMARK CE. Chronic pancreatitis and small intestinal bacterial overgrowth[J]. Pancreatology, 2018, 18( 4): 360- 362. DOI: 10.1016/j.pan.2018.04.011. [18] THERRIEN A, BOUCHARD S, SIDANI S, et al. Prevalence of small intestinal bacterial overgrowth among chronic pancreatitis patients: A case-control study[J]. Can J Gastroenterol Hepatol, 2016, 2016: 7424831. DOI: 10.1155/2016/7424831. [19] NÍ CHONCHUBHAIR HM, BASHIR Y, DOBSON M, et al. The prevalence of small intestinal bacterial overgrowth in non-surgical patients with chronic pancreatitis and pancreatic exocrine insufficiency(PEI)[J]. Pancreatology, 2018, 18( 4): 379- 385. DOI: 10.1016/j.pan.2018.02.010. [20] LEE AA, BAKER JR, WAMSTEKER EJ, et al. Small intestinal bacterial overgrowth is common in chronic pancreatitis and associates with diabetes, chronic pancreatitis severity, low zinc levels, and opiate use[J]. Am J Gastroenterol, 2019, 114( 7): 1163- 1171. DOI: 10.14309/ajg.0000-000000000200. [21] PAN LL, LI BB, PAN XH, et al. Gut microbiota in pancreatic diseases: Possible new therapeutic strategies[J]. Acta Pharmacol Sin, 2021, 42( 7): 1027- 1039. DOI: 10.1038/s41401-020-00532-0. [22] SUN J, FURIO L, MECHERI R, et al. Pancreatic β-cells limit autoimmune diabetes via an immunoregulatory antimicrobial peptide expressed under the influence of the gut microbiota[J]. Immunity, 2015, 43( 2): 304- 317. DOI: 10.1016/j.immuni.2015.07.013. [23] O’DWYER DN, ASHLEY SL, GURCZYNSKI SJ, et al. Lung microbiota contribute to pulmonary inflammation and disease progression in pulmonary fibrosis[J]. Am J Respir Crit Care Med, 2019, 199( 9): 1127- 1138. DOI: 10.1164/rccm.201809-1650OC. [24] NEEDELL JC, IR D, ROBERTSON CE, et al. Maternal treatment with short-chain fatty acids modulates the intestinal microbiota and immunity and ameliorates type 1 diabetes in the offspring[J]. PLoS One, 2017, 12( 9): e0183786. DOI: 10.1371/journal.pone.0183786. [25] YANG L, LIN HQ, LIN WT, et al. Exercise ameliorates insulin resistance of type 2 diabetes through motivating short-chain fatty acid-mediated skeletal muscle cell autophagy[J]. Biology, 2020, 9( 8): 203. DOI: 10.3390/biology9080203. [26] ZHU XZ, ZHANG HH, MENG XY, et al. Short-chain fatty acids reduced insulin resistance and pancreatic damage in type 2 diabetic mice[J]. Mod Food Sci Technol, 2020, 36( 8): 1- 7. DOI: 10.13982/j.mfst.1673-9078.2020.8.0146.朱晓振, 张菡菡, 孟现尧, 等. 短链脂肪酸改善2型糖尿病小鼠胰岛素抵抗和胰腺损伤[J]. 现代食品科技, 2020, 36( 8): 1- 7. DOI: 10.13982/j.mfst.1673-9078.2020.8.0146. [27] SHERMAN MH. Stellate cells in tissue repair, inflammation, and cancer[J]. Annu Rev Cell Dev Biol, 2018, 34: 333- 355. DOI: 10.1146/annurev-cellbio-100617-062855. [28] GRYSHCHENKO O, GERASIMENKO JV, GERASIMENKO OV, et al. Ca2+ signals mediated by bradykinin type 2 receptors in normal pancreatic stellate cells can be inhibited by specific Ca2+ channel blockade[J]. J Physiol, 2016, 594( 2): 281- 293. DOI: 10.1113/JP271468. [29] TIAN L, LU ZP, CAI BB, et al. Activation of pancreatic stellate cells involves an EMT-like process[J]. Int J Oncol, 2016, 48( 2): 783- 792. DOI: 10.3892/ijo.2015.3282. [30] XIANG XH, XIA SH. Microenvironment of pancreatic stellate cells in chronic pancreatitis[J]. World Chin J Dig, 2017, 25( 28): 2518- 2527. DOI: 10.11569/wcjd.v25.28.2518.向晓辉, 夏时海. 胰腺星状细胞微环境与慢性胰腺炎[J]. 世界华人消化杂志, 2017, 25( 28): 2518- 2527. DOI: 10.11569/wcjd.v25.28.2518. [31] MEI D, ZHANG JZ, LIANG XQ, et al. Role of cytokines in pancreatic fibrosis of chronic pancreatitis and intervention of traditional Chinese medicine[J]. Chin J Surg Integr Tradit West Med, 2016, 22( 1): 97- 100. DOI: 10.3969/j.issn.1007-6948.2016.01.035.梅丹, 张静喆, 梁晓强, 等. 细胞因子在慢性胰腺炎胰腺纤维化中的作用及中药干预[J]. 中国中西医结合外科杂志, 2016, 22( 1): 97- 100. DOI: 10.3969/j.issn.1007-6948.2016.01.035. [32] LU HN, XU F, ZHANG QB, et al. Regulatory effects of short-chain fatty acids on oxidative stress and activation of pancreatic stellate cells[J]. Chin J Pancreatol, 2024, 24( 3): 210- 215. DOI: 10.3760/cma.j.cn115667-20240111-00012.陆宏娜, 许丰, 张秋波, 等. 短链脂肪酸对胰腺星状细胞氧化应激和活化的调控作用[J]. 中华胰腺病杂志, 2024, 24( 3): 210- 215. DOI: 10.3760/cma.j.cn115667-20240111-00012. [33] LI RP. HAS2 promotes pancreatic stellate cells activation under TGF-β1/MeCP2 axis in chronic pancreatitis[D]. Suzhou: Soochow University, 2023. DOI: 10.27351/d.cnki.gszhu.2023.002672.李瑞平. HAS2受TGF-β1/MeCP2轴作用调控慢性胰腺炎纤维化的机制研究[D]. 苏州: 苏州大学, 2023. DOI: 10.27351/d.cnki.gszhu.2023.002672. [34] ZHU YZ, ZHU C, ZHOU S, et al. Effect of pancreatic stellate cell activation regulated by microRNA on fibrosis of chronic pancreatitis[J]. J Clin Hepatol, 2017, 33( 8): 1603- 1607. DOI: 10.3969/j.issn.1001-5256.2017.08.042.朱奕舟, 朱昌, 周姝, 等. microRNA调控胰腺星状细胞活化对慢性胰腺炎纤维化进程的影响[J]. 临床肝胆病杂志, 2017, 33( 8): 1603- 1607. DOI: 10.3969/j.issn.1001-5256.2017.08.042. [35] NI JB, HU GY, WAN R, et al. Involvement of interleukin-17A in caerulein-induced chronic pancreatitis in mice[J]. Prog Mod Biomed, 2017, 17( 5): 801- 805. DOI: 10.13241/j.cnki.pmb.2017.05.001.倪建波, 胡国勇, 万荣, 等. 白介素-17A在小鼠慢性胰腺炎中的作用研究[J]. 现代生物医学进展, 2017, 17( 5): 801- 805. DOI: 10.13241/j.cnki.pmb.2017.05.001. [36] ZHENG MF, LI HY, SUN L, et al. Interleukin-6 participates in human pancreatic stellate cell activation and collagen I production via TGF-β1/Smad pathway[J]. Cytokine, 2021, 143: 155536. DOI: 10.1016/j.cyto.2021.155536. [37] KOMAR HM, SERPA G, KERSCHER C, et al. Inhibition of Jak/STAT signaling reduces the activation of pancreatic stellate cells in vitro and limits caerulein-induced chronic pancreatitis in vivo[J]. Sci Rep, 2017, 7( 1): 1787. DOI: 10.1038/s41598-017-01973-0. [38] MANOHAR M, KANDIKATTU HK, VERMA AK, et al. IL-15 regulates fibrosis and inflammation in a mouse model of chronic pancreatitis[J]. Am J Physiol Gastrointest Liver Physiol, 2018, 315( 6): G954- G965. DOI: 10.1152/ajpgi.00139.2018. [39] KANDIKATTU HK, MANOHAR M, UPPARAHALLI VENKATESHAIAH S, et al. Chronic inflammation promotes epithelial-mesenchymal transition-mediated malignant phenotypes and lung injury in experimentally-induced pancreatitis[J]. Life Sci, 2021, 278: 119640. DOI: 10.1016/j.lfs.2021.119640. [40] COBO F, RODRÍGUEZ-GRANGER J, SAMPEDRO A, et al. Infected breast cyst due to Prevotella buccae resistant to metronidazole[J]. Anaerobe, 2017, 48: 177- 178. DOI: 10.1016/j.anaerobe.2017.08.015. [41] KOUCHAKI E, TAMTAJI OR, SALAMI M, et al. Clinical and metabolic response to probiotic supplementation in patients with multiple sclerosis: A randomized, double-blind, placebo-controlled trial[J]. Clin Nutr, 2017, 36( 5): 1245- 1249. DOI: 10.1016/j.clnu.2016.08.015. [42] WANG W, XIAO Y, WANG XQ, et al. Disordered gut microbiota in children who have chronic pancreatitis and different functional gene mutations[J]. Clin Transl Gastroenterol, 2020, 11( 3): e00150. DOI: 10.14309/ctg.0000000000000150. [43] RAMMOHAN A, SATHYANESAN J, RAJENDRAN K, et al. Synbiotics in surgery for chronic pancreatitis: Are they truly effective? A single-blind prospective randomized control trial[J]. Ann Surg, 2015, 262( 1): 31- 37. DOI: 10.1097/SLA.0000000000001077. [44] ZAFAR H, JIMENEZ B, SCHNEIDER A. Small intestinal bacterial overgrowth: Current update[J]. Curr Opin Gastroenterol, 2023, 39( 6): 522- 528. DOI: 10.1097/MOG.0000000000000971. [45] ASLAN I, TARHAN CELEBI L, KAYHAN H, et al. Probiotic formulations containing fixed and essential oils ameliorates SIBO-induced gut dysbiosis in rats[J]. Pharmaceuticals, 2023, 16( 7): 1041. DOI: 10.3390/ph16-071041. [46] HUANG YW. The protective effect and mechanism of Akkermansia muciniphila on chronic pancreatitis[D]. Wuxi: Jiangnan University, 2021. DOI: 10.27169/d.cnki.gwqgu.2021.000231.黄伊雯. 新型益生菌Akkermansia muciniphila对慢性胰腺炎的作用及机制研究[D]. 无锡: 江南大学, 2021. DOI: 10.27169/d.cnki.gwqgu.2021.000231. [47] BAKKER GJ, NIEUWDORP M. Fecal microbiota transplantation: Therapeutic potential for a multitude of diseases beyond Clostridium difficile[J]. Microbiol Spectr, 2017, 5( 4). DOI: 10.1128/microbiolspec.BAD-0008-2017. [48] WORTELBOER K, NIEUWDORP M, HERREMA H. Fecal microbiota transplantation beyond Clostridioides difficile infections[J]. EBioMedicine, 2019, 44: 716- 729. DOI: 10.1016/j.ebiom.2019.05.066. [49] HE YF, WANG JC, WU FF, et al. Research progress in fecal microbiota transplantation in hepatobiliary and pancreatic surgery[J/OL]. Chin J Hepat Surg(Electronic Edition), 2025, 14( 4): 646- 653. DOI: 10.3877/cma.j.issn.2095-3232.2025.04.023.何勇飞, 王继才, 吴芬芳, 等. 粪菌移植在肝胆胰外科领域研究进展[J/OL]. 中华肝脏外科手术学电子杂志, 2025, 14( 4): 646- 653. DOI: 10.3877/cma.j.issn.2095-3232.2025.04.023. [50] WANG JW, KUO CH, KUO FC, et al. Fecal microbiota transplantation: Review and update[J]. J Formos Med Assoc, 2019, 118: S23- S31. DOI: 10.1016/j.jfma.2018.08.011. [51] QURAISHI MN, WIDLAK M, BHALA N, et al. Systematic review with meta-analysis: The efficacy of faecal microbiota transplantation for the treatment of recurrent and refractory Clostridium difficile infection[J]. Aliment Pharmacol Ther, 2017, 46( 5): 479- 493. DOI: 10.1111/apt.14201. [52] LI YT, CAI HF, WANG ZH, et al. Systematic review with meta-analysis: Long-term outcomes of faecal microbiota transplantation for Clostridium difficile infection[J]. Aliment Pharmacol Ther, 2016, 43( 4): 445- 457. DOI: 10.1111/apt.13492. [53] CHENG SS, MA X, GENG SJ, et al. Fecal microbiota transplantation beneficially regulates intestinal mucosal autophagy and alleviates gut barrier injury[J]. mSystems, 2018, 3( 5): e00137-18. DOI: 10.1128/mSystems.00137-18. -



 PDF下载 ( 1035 KB)
PDF下载 ( 1035 KB)

 下载:
下载:


