胰岛素样生长因子-1/胰岛素样生长因子-1受体信号通路调控肝纤维化的作用机制
DOI: 10.12449/JCH260227
The mechanism of action of the insulin-like growth factor-1/insulin-like growth factor-1 receptor signaling pathway in regulating liver fibrosis
-
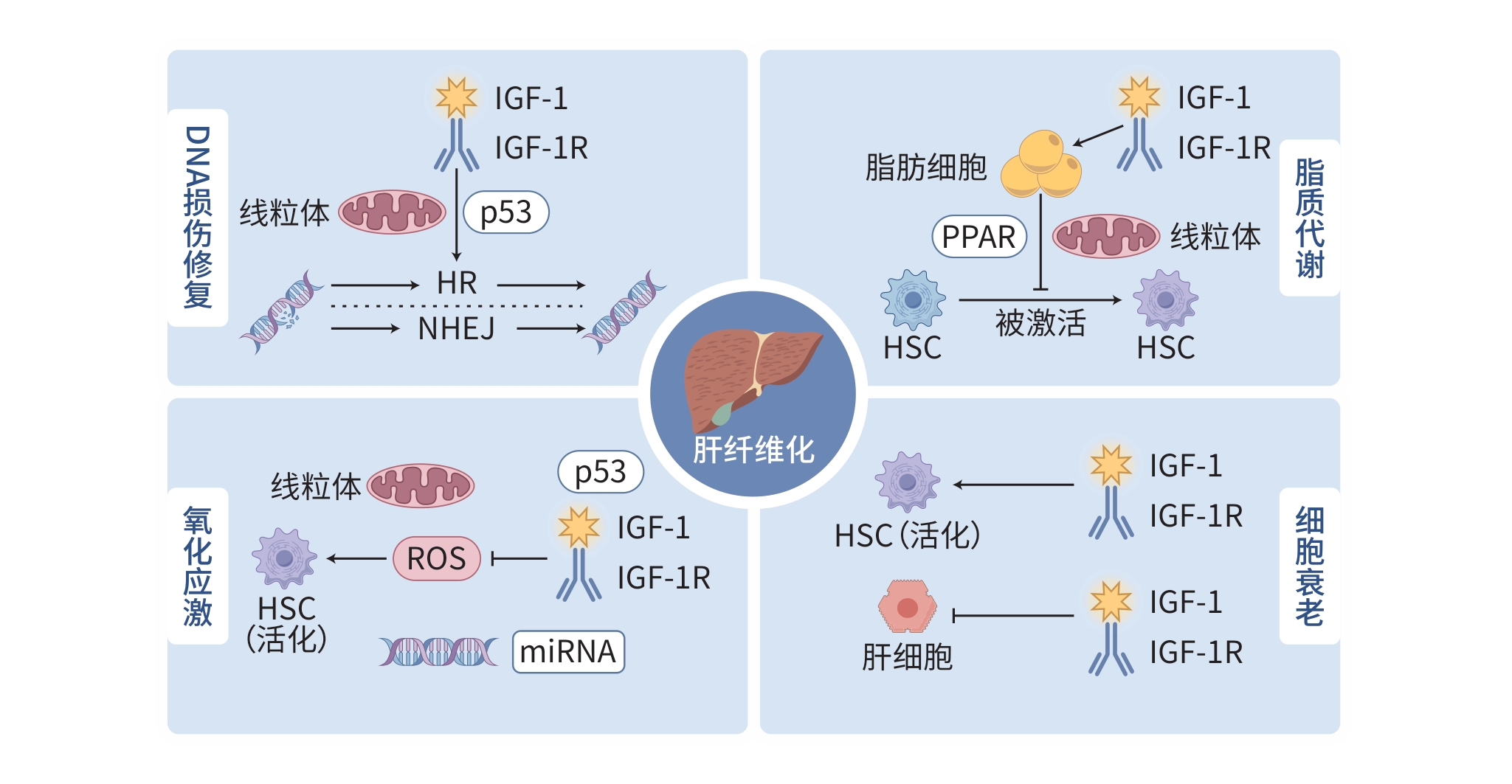
摘要: 肝纤维化由病毒感染、酒精摄入和代谢相关损伤等多种因素引起,导致正常组织被纤维瘢痕替代。胰岛素样生长因子1(IGF-1)作为一种细胞增殖调控因子,通过与其受体(IGF-1R)结合,参与调控细胞周期、促进细胞增殖与分化,并抑制细胞凋亡。研究表明,IGF-1/IGF-1R信号通路可通过影响肝细胞的衰老和凋亡、肝星状细胞的活化与增殖以及内皮细胞的功能失调等调控肝纤维化进程。此外,IGF-1/IGF-1R信号系统还可调控DNA损伤修复、细胞增殖、脂质代谢、细胞衰老及氧化应激等多种机制,为肝纤维化的预防和治疗提供了新的策略与潜在靶点。本文总结了IGF-1/IGF-1R及其信号转导系统在不同细胞中通过调控DNA损伤修复介导肝纤维化的作用机制,以期为肝纤维化的治疗提供理论依据。Abstract: Liver fibrosis is caused by various factors such as viral infection, alcohol intake, and metabolism-related damage, leading to the replacement of normal tissue by fibrous scars. As a regulatory factor for cell proliferation, insulin-like growth factor 1 (IGF-1) participates in the regulation of cell cycle, the promotion of cell proliferation and differentiation, and the inhibition of cell apoptosis by binding to its receptor insulin-like growth factor-1 receptor (IGF-1R). Studies have shown that the IGF-1/IGF-1R signaling pathway can regulate the process of liver fibrosis by affecting the senescence and apoptosis of hepatocytes, the activation and proliferation of hepatic stellate cells, and the dysfunction of endothelial cells. In addition, the IGF-1/IGF-1R signaling system can also regulate multiple mechanisms such as DNA damage repair, cell proliferation, lipid metabolism, cell senescence, and oxidative stress, thereby providing new strategies and potential targets for the prevention and treatment of liver fibrosis. This article summarizes the mechanism of action of IGF-1/IGF-1R and its signal transduction system in mediating liver fibrosis by regulating DNA damage repair in different cells, in order to provide a theoretical basis for the treatment of liver fibrosis.
-
Key words:
- Insulin-Like Growth Factor I /
- Hepatic Fibrosis /
- DNA Damage /
- Signal Transduction
-
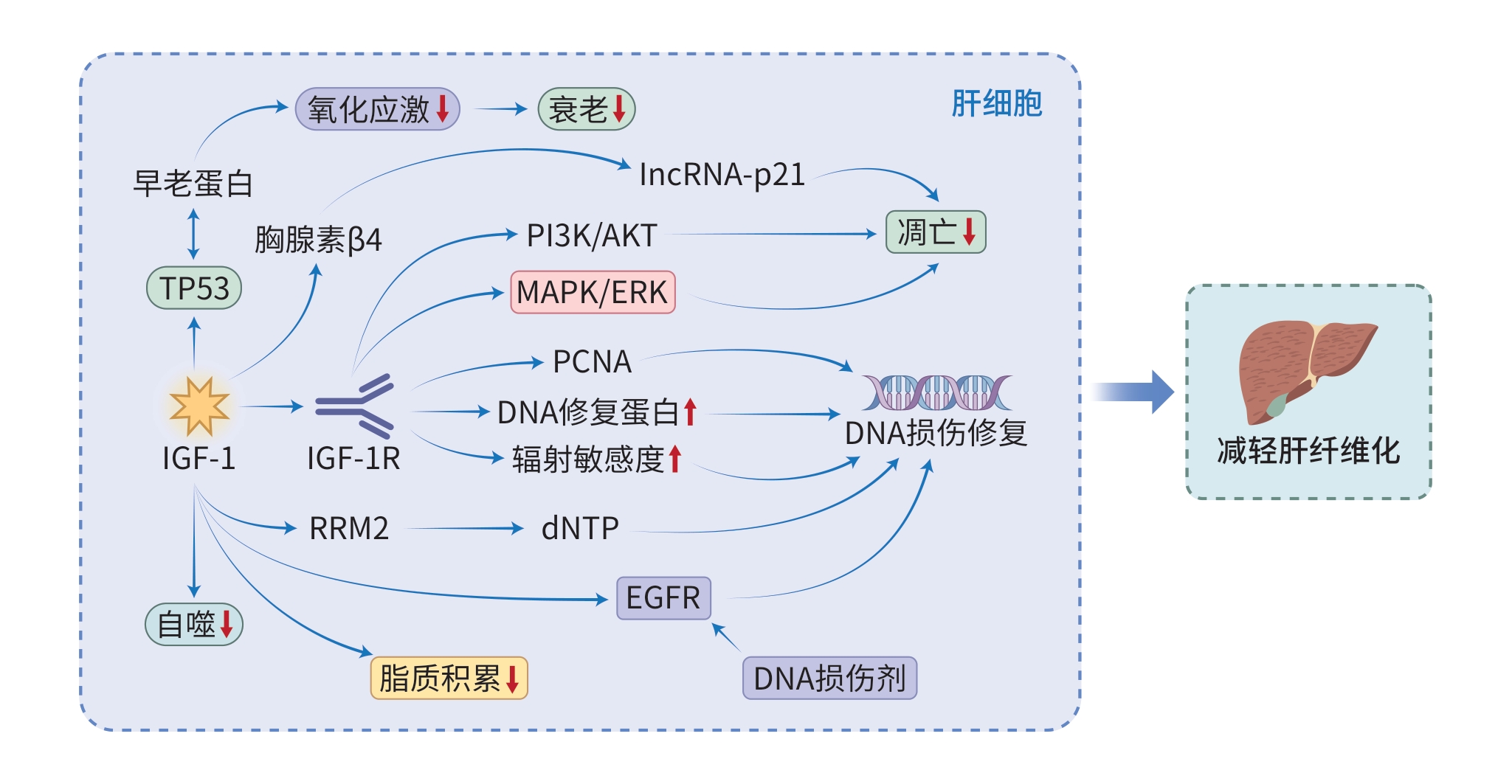
注: IGF-1,胰岛素样生长因子1;IGF-1R,胰岛素样生长因子1受体;lncRNA-p21,长链非编码RNAp21; PI3K,磷脂酰肌醇3激酶;AKT,蛋白激酶B;MAPK,丝裂原激活的蛋白激酶;ERK,胞外信号调节激酶;PCNA,增殖细胞核抗原;RRM2, 核糖核苷酸还原酶M2 亚基; dNTP, 脱氧核糖核苷三磷酸; EGFR, 表皮生长因子受体。
图 2 IGF-1/IGF-1R轴在肝细胞中介导肝纤维化的机制
Figure 2. The mechanism by which the IGF-1/IGF-1R axis mediates hepatic fibrosis in hepatocytes
-
[1] HALLSWORTH K, MCPHERSON S, ANSTEE QM, et al. Digital intervention with lifestyle coach support to target dietary and physical activity behaviors of adults with nonalcoholic fatty liver disease: Systematic development process of VITALISE using intervention mapping[J]. J Med Internet Res, 2021, 23( 1): e20491. DOI: 10.2196/20491. [2] DAI WM, LU LG, CAI XB. Association between liver sinusoidal endothelial cells and liver fibrosis[J]. J Clin Hepatol, 2023, 39( 2): 419- 423. DOI: 10.3969/j.issn.1001-5256.2023.02.027.戴伟明, 陆伦根, 蔡晓波. 肝窦内皮细胞与肝纤维化的关系[J]. 临床肝胆病杂志, 2023, 39( 2): 419- 423. DOI: 10.3969/j.issn.1001-5256.2023.02.027. [3] ZHAO HB, JIN H, XIAN JF, et al. Effect of ketogenic diets on body composition and metabolic parameters of cancer patients: A systematic review and meta-analysis[J]. Nutrients, 2022, 14( 19): 4192. DOI: 10.3390/nu14194192. [4] KRUGER DT, ALEXI X, OPDAM M, et al. IGF-1R pathway activation as putative biomarker for linsitinib therapy to revert tamoxifen resistance in ER-positive breast cancer[J]. Int J Cancer, 2020, 146( 8): 2348- 2359. DOI: 10.1002/ijc.32668. [5] LUO XY, JIANG XK, LI J, et al. Insulin-like growth factor-1 attenuates oxidative stress-induced hepatocyte premature senescence in liver fibrogenesis via regulating nuclear p53-progerin interaction[J]. Cell Death Dis, 2019, 10( 6): 451. DOI: 10.1038/s41419-019-1670-6. [6] TANG WY, ZHANG M, WANG Y, et al. IGF-1 inhibits palmitic acid-induced mitochondrial apoptosis in macrophages[J]. Mol Med Rep, 2023, 28( 6): 234. DOI: 10.3892/mmr.2023.13121. [7] ZHU YH, CHEN L, SONG BY, et al. Insulin-like growth factor-2(IGF-2) in fibrosis[J]. Biomolecules, 2022, 12( 11): 1557. DOI: 10.3390/biom-12111557. [8] MOREAU F, KIRK NS, ZHANG F, et al. Interaction of a viral insulin-like peptide with the IGF-1 receptor produces a natural antagonist[J]. Nat Commun, 2022, 13( 1): 6700. DOI: 10.1038/s41467-022-34391-6. [9] WERNER H. The IGF1 signaling pathway: From basic concepts to therapeutic opportunities[J]. Int J Mol Sci, 2023, 24( 19): 14882. DOI: 10.3390/ijms241914882. [10] JÓZEFIAK A, LARSKA M, POMORSKA-MÓL M, et al. The IGF-1 signaling pathway in viral infections[J]. Viruses, 2021, 13( 8): 1488. DOI: 10.3390/v13081488. [11] SHANMUGALINGAM T, BOSCO C, RIDLEY AJ, et al. Is there a role for IGF-1 in the development of second primary cancers[J]. Cancer Med, 2016, 5( 11): 3353- 3367. DOI: 10.1002/cam4.871. [12] GARCÍA-MATO Á, CERVANTES B, RODRÍGUEZ-DE LA ROSA L, et al. IGF-1 controls metabolic homeostasis and survival in HEI-OC1 auditory cells through AKT and mTOR signaling[J]. Antioxidants(Basel), 2023, 12( 2): 233. DOI: 10.3390/antiox12020233. [13] JIANG Y, WANG JQ. Research progress of DNA methylation in liver fibrosis[J]. Chin Hepatol, 2024, 29( 4): 468- 471. DOI: 10.3969/j.issn.1008-1704.2024.04.024.蒋月, 王建青. DNA甲基化在肝纤维化中的研究进展[J]. 肝脏, 2024, 29( 4): 468- 471. DOI: 10.3969/j.issn.1008-1704.2024.04.024. [14] CHO SS, LEE JH, KIM KM, et al. REDD1 attenuates hepatic stellate cell activation and liver fibrosis via inhibiting of TGF-β/Smad signaling pathway[J]. Free Radic Biol Med, 2021, 176: 246- 256. DOI: 10.1016/j.freeradbiomed.2021.10.002. [15] YU SY, JI G, ZHANG L. The role of p53 in liver fibrosis[J]. Front Pharmacol, 2022, 13: 1057829. DOI: 10.3389/fphar.2022.1057829. [16] CARUSILLO A, MUSSOLINO C. DNA damage: From threat to treatment[J]. Cells, 2020, 9( 7): 1665. DOI: 10.3390/cells9071665. [17] HUO SM, LI B, DU JY, et al. Dibutyl phthalate induces liver fibrosis via p38MAPK/NF-κB/NLRP3-mediated pyroptosis[J]. Sci Total Environ, 2023, 897: 165500. DOI: 10.1016/j.scitotenv.2023.165500. [18] LIN G, LI WB, HONG WB, et al. Spinosin inhibits activated hepatic stellate cell to attenuate liver fibrosis by targeting Nur77/ASK1/p38 MAPK signaling pathway[J]. Eur J Pharmacol, 2024, 966: 176270. DOI: 10.1016/j.ejphar.2023.176270. [19] LI M, YANG ZF, SONG ZY, et al. ATM deficiency aggravates the progression of liver fibrosis induced by carbon tetrachloride in mice[J]. Toxicology, 2023, 484: 153397. DOI: 10.1016/j.tox.2022.153397. [20] ZOU L, SHI CF, WANG DW, et al. Long non-coding RNA-non-coding RNA activated by DNA damage inhibition suppresses hepatic stellate cell activation via microRNA-495-3p/sphingosine 1-phosphate receptor 3 axis[J]. Bioengineered, 2022, 13( 3): 6150- 6162. DOI: 10.1080/21655979.2022.2037841. [21] YANG L, FU WL, ZHU Y, et al. Tβ4 suppresses lincRNA-p21-mediated hepatic apoptosis and fibrosis by inhibiting PI3K-AKT-NF-κB pathway[J]. Gene, 2020, 758: 144946. DOI: 10.1016/j.gene.2020.144946. [22] ZHANG XF, WU XW, HU QY, et al. Mitochondrial DNA in liver inflammation and oxidative stress[J]. Life Sci, 2019, 236: 116464. DOI: 10.1016/j.lfs.2019.05.020. [23] MELIN N, YARAHMADOV T, SANCHEZ-TALTAVULL D, et al. A new mouse model of radiation-induced liver disease reveals mitochondrial dysfunction as an underlying fibrotic stimulus[J]. JHEP Rep, 2022, 4( 7): 100508. DOI: 10.1016/j.jhepr.2022.100508. [24] GUAN X, YAN QY, WANG DD, et al. IGF-1 signaling regulates mitochondrial remodeling during myogenic differentiation[J]. Nutrients, 2022, 14( 6): 1249. DOI: 10.3390/nu14061249. [25] HOU XW, LI ZH, HIGASHI Y, et al. Insulin-like growth factor I prevents cellular aging via activation of mitophagy[J]. J Aging Res, 2020, 2020: 4939310. DOI: 10.1155/2020/4939310. [26] NISHIZAWA H, IGUCHI G, FUKUOKA H, et al. IGF-I induces senescence of hepatic stellate cells and limits fibrosis in a p53-dependent manner[J]. Sci Rep, 2016, 6: 34605. DOI: 10.1038/srep34605. [27] TAKAHASHI Y. The role of growth hormone and insulin-like growth factor-I in the liver[J]. Int J Mol Sci, 2017, 18( 7): 1447. DOI: 10.3390/ijms18071447. [28] XIU AY, DING Q, LI Z, et al. Doxazosin attenuates liver fibrosis by inhibiting autophagy in hepatic stellate cells via activation of the PI3K/Akt/mTOR signaling pathway[J]. Drug Des Devel Ther, 2021, 15: 3643- 3659. DOI: 10.2147/DDDT.S317701. [29] CHENG YY, LI WQ, GUI RR, et al. Dual characters of GH-IGF1 signaling pathways in radiotherapy and post-radiotherapy repair of cancers[J]. Front Cell Dev Biol, 2021, 9: 671247. DOI: 10.3389/fcell.2021.671247. [30] CHESNOKOVA V, MELMED S. Peptide hormone regulation of DNA damage responses[J]. Endocr Rev, 2020, 41( 4): bnaa009. DOI: 10.1210/endrev/bnaa009. [31] RIEUNIER G, WU XN, HARRIS LE, et al. Targeting IGF perturbs global replication through ribonucleotide reductase dysfunction[J]. Cancer Res, 2021, 81( 8): 2128- 2141. DOI: 10.1158/0008-5472.CAN-20-2860. [32] POREBA E, DURZYNSKA J. Nuclear localization and actions of the insulin-like growth factor 1(IGF-1) system components: Transcriptional regulation and DNA damage response[J]. Mutat Res Rev Mutat Res, 2020, 784: 108307. DOI: 10.1016/j.mrrev.2020.108307. [33] TANG T, JIANG G, SHAO J, et al. lncRNA MSTRG4710 promotes the proliferation and differentiation of preadipocytes through miR-29b-3p/IGF1 axis[J]. Int J Mol Sci, 2023, 24( 21): 15715. DOI: 10.3390/ijms24-2115715. [34] FIORE E, DOMÍNGUEZ LM, BAYO J, et al. Human umbilical cord perivascular cells-derived extracellular vesicles mediate the transfer of IGF-I to the liver and ameliorate hepatic fibrogenesis in mice[J]. Gene Ther, 2020, 27( 1-2): 62- 73. DOI: 10.1038/s41434-019-0102-7. [35] CHEN SH, GAO JL, WANG LP, et al. The IGF1 system is involved in radiation-induced activation of hepatic stellate cells[C]// The 10th National Toxicology Congress of Chinese Society of Toxicology, 2023: 253.陈思含, 高嘉璐, 王路平, 等. IGF1系统参与辐射诱导的HSC的激活[C]// 中国毒理学会第十次全国毒理学大会, 2023: 253. [36] GUI RR, LI WQ, LI ZP, et al. Effects and potential mechanisms of IGF1/IGF1R in the liver fibrosis: A review[J]. Int J Biol Macromol, 2023, 251: 126263. DOI: 10.1016/j.ijbiomac.2023.126263. [37] WANG YF, WANG PY, YU YB, et al. Hepatocyte Ninjurin2 promotes hepatic stellate cell activation and liver fibrosis through the IGF1R/EGR1/PDGF-BB signaling pathway[J]. Metabolism, 2023, 140: 155380. DOI: 10.1016/j.metabol.2022.155380. [38] LIU D, WU ZG, GAO JH, et al. CircUTRN24/miR-483-3p/IGF-1 regulates autophagy mediated liver fibrosis in biliary atresia[J]. Mol Biotechnol, 2024, 66( 6): 1424- 1433. DOI: 10.1007/s12033-023-00802-2. [39] NEWSOME PN, AMBERY P. Incretins(GLP-1 receptor agonists and dual/triple agonists) and the liver[J]. J Hepatol, 2023, 79( 6): 1557- 1565. DOI: 10.1016/j.jhep.2023.07.033. [40] ZHANG H, WANG XS, HU B, et al. Human umbilical cord mesenchymal stem cells attenuate diabetic nephropathy through the IGF1R-CHK2-p53 signalling axis in male rats with type 2 diabetes mellitus[J]. J Zhejiang Univ Sci B, 2024, 25( 7): 568- 580. DOI: 10.1631/jzus.B2300182. [41] GUSSCOTT S, JENKINS CE, LAM SH, et al. IGF1R derived PI3K/AKT signaling maintains growth in a subset of human T-cell acute lymphoblastic leukemias[J]. PLoS One, 2016, 11( 8): e0161158. DOI: 10.1371/journal.pone.0161158. [42] OCK S, HAM W, KANG CW, et al. IGF-1 protects against angiotensin II-induced cardiac fibrosis by targeting αSMA[J]. Cell Death Dis, 2021, 12( 7): 688. DOI: 10.1038/s41419-021-03965-5. [43] TIE Y, TANG F, PENG DD, et al. TGF-beta signal transduction: Biology, function and therapy for diseases[J]. Mol Biomed, 2022, 3( 1): 45. DOI: 10.1186/s43556-022-00109-9. [44] PIÑEIRO-HERMIDA S, LÓPEZ IP, ALFARO-ARNEDO E, et al. IGF1R deficiency attenuates acute inflammatory response in a bleomycin-induced lung injury mouse model[J]. Sci Rep, 2017, 7( 1): 4290. DOI: 10.1038/s41598-017-04561-4. [45] NEVOLA R, EPIFANI R, IMBRIANI S, et al. GLP-1 receptor agonists in non-alcoholic fatty liver disease: Current evidence and future perspectives[J]. Int J Mol Sci, 2023, 24( 2): 1703. DOI: 10.3390/ijms24-021703. [46] ZHU T, PAN MJ, DING QN, et al. Targeting insulin-like growth factor 1 signaling with Linsitinib to modulate fibroblast plasticity and attenuate pulmonary fibrosis[J]. Int Immunopharmacol, 2025, 161: 115084. DOI: 10.1016/j.intimp.2025.115084. [47] MA Z, XIE KH, XUE XY, et al. Si-Wu-Tang attenuates hepatocyte PANoptosis and M1 polarization of macrophages in non-alcoholic fatty liver disease by influencing the intercellular transfer of mtDNA[J]. J Ethnopharmacol, 2024, 328: 118057. DOI: 10.1016/j.jep.2024.118057. [48] TIAN MX, LIU WL, YOU HJ, et al. Protective effect of Yiguanjian decoction against DNA damage on concanavalin A-induced liver injury mice model[J]. J Tradit Chin Med, 2016, 36( 4): 471- 478. DOI: 10.1016/s0254-6272(16)30064-4. [49] OUYANG H, MIAO H, LI Z, et al. Yinhuang granule alleviates carbon tetrachloride-induced liver fibrosis in mice and its mechanism[J]. World J Hepatol, 2024, 16( 2): 264- 278. DOI: 10.4254/wjh.v16.i2.264. [50] JIN H, LIAN N, ZHANG F, et al. Activation of PPARγ/P53 signaling is required for curcumin to induce hepatic stellate cell senescence[J]. Cell Death Dis, 2016, 7( 4): e2189. DOI: 10.1038/cddis.2016.92. [51] LI YJ, LIU RP, DING MN, et al. Tetramethylpyrazine prevents liver fibrotic injury in mice by targeting hepatocyte-derived and mitochondrial DNA-enriched extracellular vesicles[J]. Acta Pharmacol Sin, 2022, 43( 8): 2026- 2041. DOI: 10.1038/s41401-021-00843-w. [52] LIU GF, WEI C, YUAN SY, et al. Wogonoside attenuates liver fibrosis by triggering hepatic stellate cell ferroptosis through SOCS1/P53/SLC7A11 pathway[J]. Phytother Res, 2022, 36( 11): 4230- 4243. DOI: 10.1002/ptr.7558. [53] LI N, ZHU CH, FU RZ, et al. Ginsenoside Rg5 inhibits lipid accumulation and hepatocyte apoptosis via the Notch1 signaling pathway in NASH mice[J]. Phytomedicine, 2024, 124: 155287. DOI: 10.1016/j.phymed.2023.155287. -



 PDF下载 ( 976 KB)
PDF下载 ( 976 KB)

 下载:
下载: